April is here, and we’re excited to share the latest research, updates, and innovations happening at NeuroNexus.
Electrochemical Impedance Spectroscopy Webinar Series
In this three-part series, Distinguished Professor Mark E. Orazem will guide attendees through the fundamentals, analysis, and real-world applications of EIS. We’re excited to collaborate with The Electrochemical Society and The International Society of Electrochemistry to bring this educational series to the community.
Introduction to EIS
📅 Wednesday, April 1, 2026 | 12 PM ET
An overview of impedance spectroscopy as a powerful electrochemical technique used across batteries, fuel cells, corrosion science, sensors, and neural electrodes. This session covers the history of EIS, key applications, and how to design measurements that maximize signal quality and meaningful data interpretation.
Interpretation, Measurement, and Analysis of EIS
📅 Wednesday, April 8, 2026 | 12 PM ET
Learn how to interpret impedance data by understanding the chemistry and physics of electrochemical systems. This session will explore measurement errors, validation using Kramers-Kronig relations, model development, and how impedance models translate into meaningful physical parameters.
Application of EIS to Neural Engineering
📅 Wednesday, April 15, 2026 | 12 PM ET
A deep dive into applying EIS to neural electrodes. Topics include measurement limitations caused by wiring and instrumentation, accuracy contour plots, and modeling electrode materials such as SIROF and TiN used in neural stimulation.

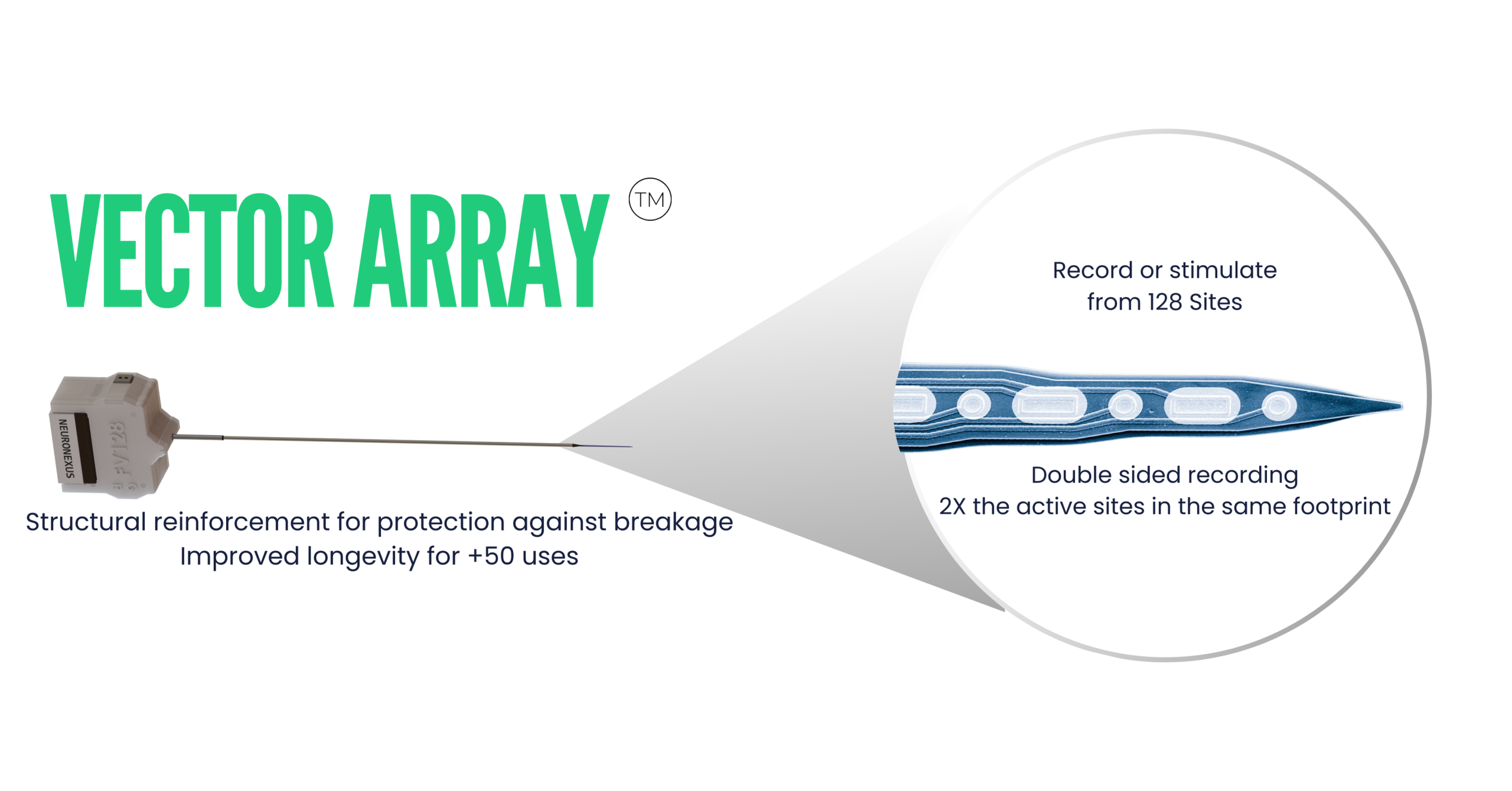
Product of the month: MINI MATRIX
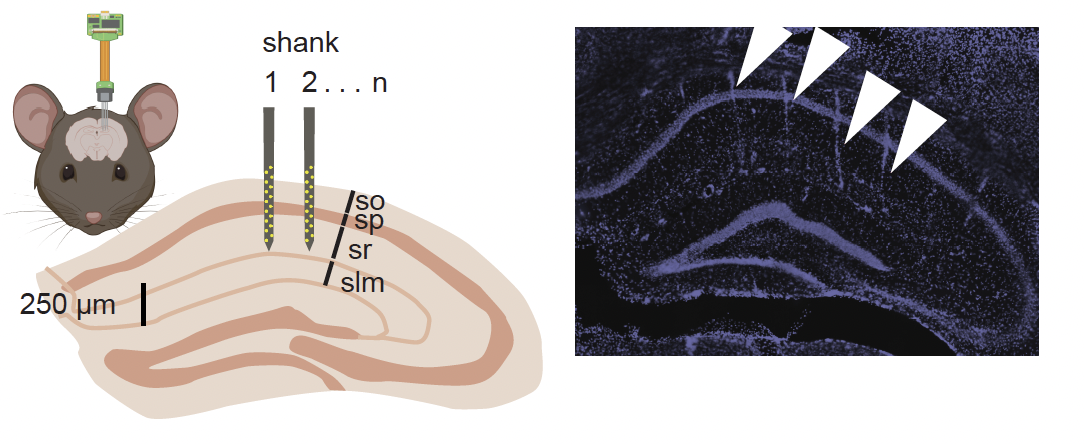
Mini Matrix is a customizable multi-shank probe engineered for high-density recordings across 3 dimensions – expanding access to insights of large populations of neurons in small animals for chronic or acute application.

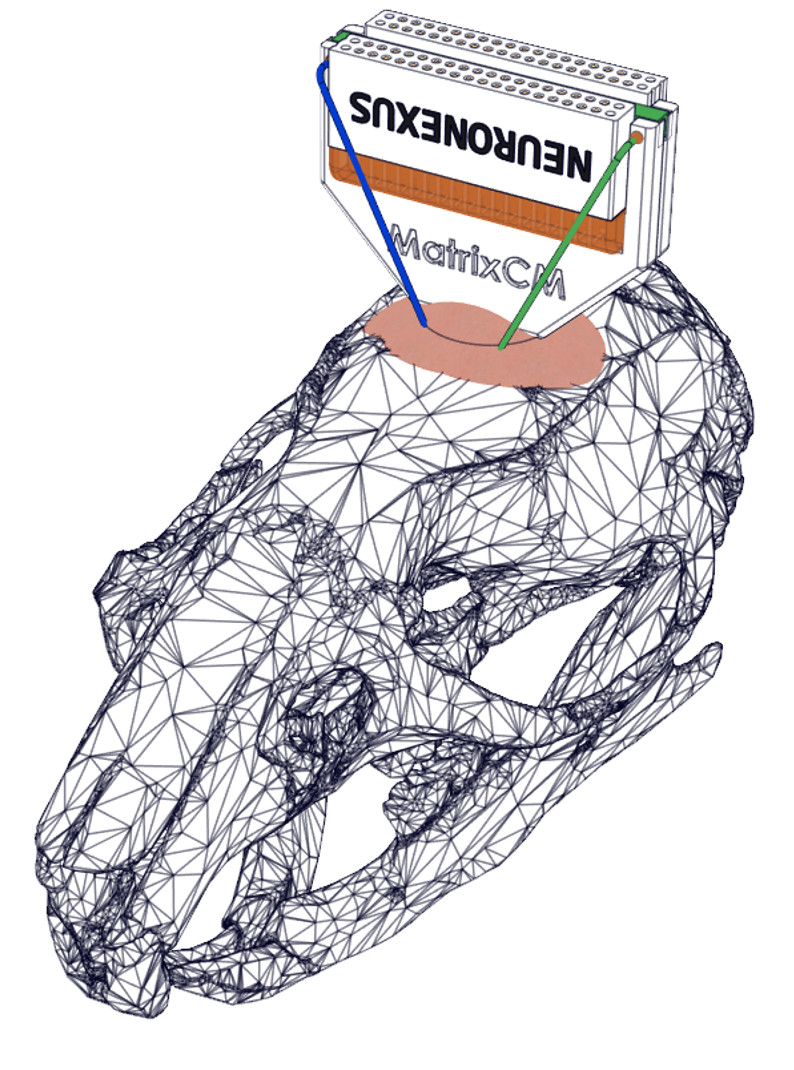

- 3D Neural Interface – The Matrix Array concurrently spans a 2D footprint and multiple depths, covering a volume of tissue and interfacing with large populations of neurons.
- High Channel Density – Record and stimulate from 32, 64, 128, or channels.
- Automated Surgical Procedure – NeuroNexus worked closely with leading primate labs to develop a low-speed, low-risk, automated implantation procedure, reducing recovery time and preserving tissue health.
- Robust – Lab-tested and refined to the smallest detail, the Matrix Array can withstand repeated acute use and demanding chronic applications.
- Customizable – Electrode length, site area, shank/site spacing, cable length, guide rod length, and headstage compatibility can all be customized for your workflow.
Learn more about the Mini Matrix
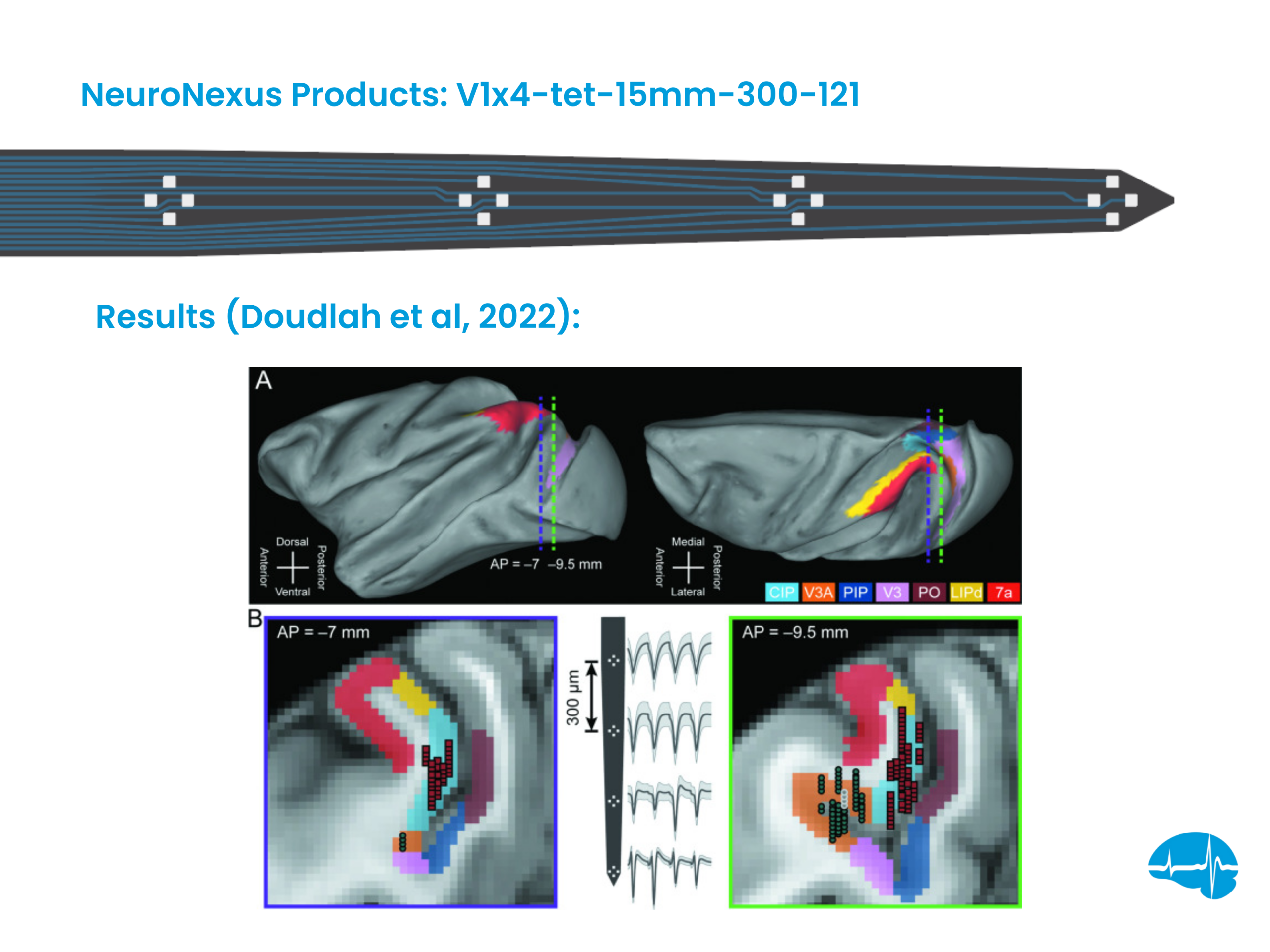
Application of the Mini Matrix
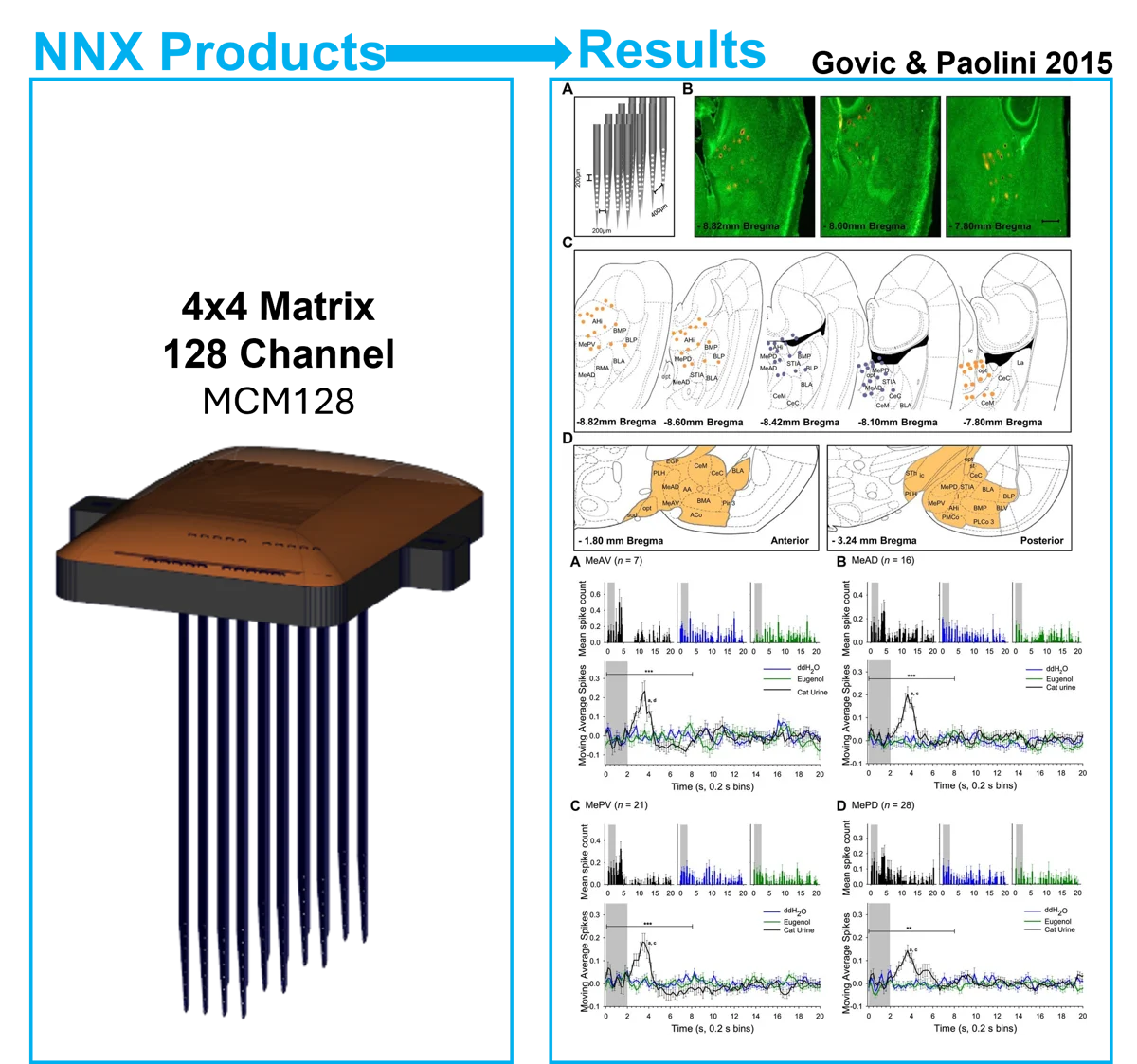
Chemosensory cues signaling predators and stimulate defensive responses in rodents. While there is a well-documented role of the amygdala in predator odor-induced fear, the nuanced physiology of heterogeneous nuclei within the amygdala remain under explored. This study analyzes defensive behaviors and amygdala electrophysiological responses to predator odor to better understand the specificity of these amygdala subnuclei.
NNx Tools Used
The team used a 128 channel Mini Matrix Array (MCM128) for electrophysiology recordings in the amygdala.
Key Findings
- The medial amygdala (MeA) demonstrated the greatest frequency of neurological responses to predator odar, followed by the basolateral and basomedial nuclei with the central amygdala having the lowest response frequency
- Information transduction occurred primarily from the cortical amygdala and MeA (ventral divisions) to other amygdala nuclei
- MeA subnuclei exhibited distinct firing patterns to predator urine, potentially revealing aspects of the underlying neurocircuitry of predator odor processing and defensiveness
Significance
These findings highlight the critical involvement of the MeA in processing olfactory cues signaling predator threat and converge with previous studies to indicate that amygdala regulation of predator odor-induced fear is restricted to a particular set of subnuclei that primarily include the MeA, particularly the ventral divisions.
More publications using Mini Matrix Arrays
The NeuroNexus Podcast
In this episode of the NeuroNexus Podcast, we sit down with ambitious postdoctoral scholar Dr. Mohamed El Tabbal from the Salk Institute for Biological Studies to talk about his innovative approach to visualizing neuronal activity. Whether you’re a long-time NeuroNexus user or just getting started in electrophysiology, you’ll find something useful, relatable, and inspiring in every conversation.